2835. 2,4-DB
Nomenclature
CAS number: 94-82-6
4-(2,4-Dichlorophenoxy)butanoic acid; 4-(2,4-dichlorophenoxy)butyric acid; Butyrac (Union Carbide); Legumex D. C10H10Cl2O3; mol wt 249.09.
C 48.22%, H 4.05%, Cl 28.47%, O 19.27%.
Description and references
Selective post-emergence, translocated herbicide. First described as a plant growth regulator: M. E. Synerholm and P. W. Zimmerman, Contrib. Boyce Thompson Inst. 14, 369 (1947). Prepn: G. W. Kitchingman, A. C. Tucker, GB 804565 and GB 883255 (1958, 1961, both to ICI). Similar in activity to 2,4-D, q.v., but more selective. Mechanism of selectivity studies: D. L. King, D. E. Bayer, Proc. West. Soc. Weed Sci. 25, 37 (1972).
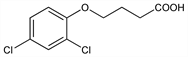
Properties
White crystals, mp 117-119°. Soly in water at 25°: 46 ppm. Sol in acetone, ethanol, diethyl ether. Slightly sol in benzene, toluene, kerosene.Derivative
Sodium salt.Nomenclature
Embutox.C10H9Cl2NaO3; mol wt 271.07.
C 44.31%, H 3.35%, Cl 26.16%, Na 8.48%, O 17.71%.
Properties
Water soluble; hard water will show precipitate as the calcium and magnesium salts.Derivative
Dimethylamine salt.Nomenclature
Butoxone.C12H17Cl2NO3; mol wt 294.17.
C 48.99%, H 5.82%, Cl 24.10%, N 4.76%, O 16.32%.