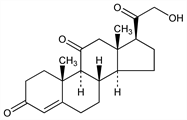
2869. 11-Dehydrocorticosterone
Nomenclature
Description and references
Found in adrenal cortex. 1000 lbs of beef glands yield 333 mg: Kendall, Cold Spring Harbor Symp. Quant. Biol. 5, 299 (1937). Isoln procedure: Mason et al., J. Biol. Chem. 114, 613 (1936); see also Kendall et al., Proc. Staff Meet. Mayo Clin. 12, 136 (1937). Prepn from corticosterone 21-acetate: Reichstein, Helv. Chim. Acta 20, 953 (1937); from desoxycholic acid: Lardon, Reichstein, ibid. 26, 747 (1943); Gallagher, Recent Progress in Hormone Research (New York, 1946) p 83; from 3α-acetoxy-11-ketobisnorcholanic acid: Sarett, J. Biol. Chem. 162, 601 (1946); J. Am. Chem. Soc. 68, 2478 (1946); from 3α-acetyl-11-ketolithocholic acid methyl ester: Wettstein, Meystre, Helv. Chim. Acta 30, 1262 (1947). Manuf: Wettstein, US 2778776 (1957 to Ciba).