2872. Dehydroergosterol
Nomenclature
CAS number: 516-85-8
(3β,22E)-Ergosta-5,7,9(11),22-tetraen-3-ol. C28H42O; mol wt 394.63.
C 85.22%, H 10.73%, O 4.05%.
Description and references
Prepd from ergosterol: Windhaus, Linsert, Ann. 465, 148 (1928); Callow, Rosenheim, J. Chem. Soc. 1933, 387. From isopyrocalciferol: Windhaus, Dimroth, Ber. 70, 376 (1937).
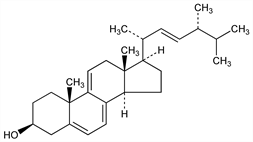
Properties
Solvated plates from alcohol, needles from ether. When dry, mp 146°. bp0.5 230°. [α]D15 +149.2° (c = 1.9 in chloroform). Absorption spectrum: Morton, de Gouveia, J. Chem. Soc. 1934, 916. One gram dissolves in 800 ml methanol. Freely sol in ether, chloroform, benzene.Derivative
Acetate.C30H44O2; mol wt 436.67.
C 82.52%, H 10.16%, O 7.33%.
Properties
mp 146°. [α]D16 +204° (c = 1.1 in chloroform).Derivative
Methyl ether.C29H44O; mol wt 408.66.
C 85.23%, H 10.85%, O 3.92%.