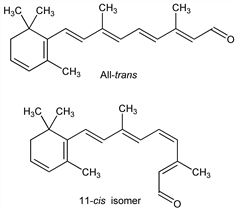
2873. 3-Dehydroretinal
Nomenclature
Description and references
Carotenoid chromophore of porphyropsin and cyanopsin, q.q.v., which are visual pigments of certain fish, crustaceans and amphibians. May also occur in combination with retinal, q.v., in mixed visual systems. Isoln from retinas of fresh-water fish: G. Wald, Nature 139, 1017 (1937). Recognition as vitamin A2 aldehyde: R. A. Morton, ibid. 153, 69 (1944). Prepn by the oxidation of vitamin A2: G. Wald, Biochim. Biophys. Acta 4, 215 (1950). Synthesis: K. R. Farrar et al., J. Chem. Soc. 1952, 1414; of stereoisomers: U. Schwieter et al., Helv. Chim. Acta 45, 517, 528, 541 (1962); R. S. H. Liu et al., J. Am. Chem. Soc. 99, 8095 (1977). HPLC separation: K. Tsukida et al., J. Chromatogr. 192, 395 (1980); T. Suzuki, M. Makino-Tasaka, Anal. Biochem. 129, 111 (1983). Biological activity: G. Wald, J. Gen. Physiol. 22, 775 (1939); idem, Fed. Proc. 12, 606 (1953). Reviews: G. Wald, Science 162, 230-239 (1968); R. Hubbard et al., Methods Enzymol. 18, 615-653 (1971). Book: The Retinoids Vol. 1-2, M. B. Sporn et al., Eds. (Academic Press, New York, 1984).