2908. d-2-Deoxyribose
Nomenclature
CAS number: 533-67-5
2-Deoxy-d-erythro-pentose; desoxyribose; d-2-deoxyarabinose; d-2-ribodesose; d-erythro-2-deoxypentose; thyminose. C5H10O4; mol wt 134.13.
C 44.77%, H 7.51%, O 47.71%.
Description and references
Isoln from deoxyribonucleic acid by acidic hydrolysis of purine deoxyribonucleosides which have been isolated by ion-exchange resin chromatography: Laland, Overend, Acta Chem. Scand. 8, 192 (1954). Synthesis: Felton, Freudenberg, J. Am. Chem. Soc. 57, 1637 (1935); Deriaz et al., J. Chem. Soc. 1949, 1879, 2836; Hough, Chem. Ind. (London) 1951, 406; Sowden, Biochem. Prep. 5, 75 (1957); I. Ziderman, E. Dimant, J. Org. Chem. 32, 1267 (1967); J. R. Hauske, H. Rapoport, ibid. 44, 2472 (1979); T. Harada, T. Mukaiyama, Chem. Lett. 1981, 1109. Review: Overend, Stacey, in Chargaff-Davidson, Nucleic Acids vol. 1, E. Chargaff, N. J. Davidson, Eds. (Academic Press, New York, 1955) pp 1-80.
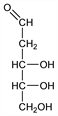
Properties
Crystals from isopropanol, mp 91°. Shows mutarotation. Final [α]D22 -56.2° (H2O). Sol in water, pyridine. Slightly sol in alc.Derivative
1,3,4-Triacetate.C11H16O7; mol wt 260.24.
C 50.77%, H 6.20%, O 43.04%.