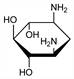
2909. 2-Deoxystreptamine
Nomenclature
Description and references
Important component of several aminocyclitol antibiotics. Production by acid hydrolysis of neamine, q.v. and structure: F. A. Kuehl et al., J. Am. Chem. Soc. 73, 881 (1952). Also obtained from hydrolysis products of kanamycins, paromomycins, gentamicins, q.q.v. and other aminoglycoside antibiotics. See K. L. Rinehart, J. Infect. Dis. 119, 345 (1969); S. Hanessian, T. H. Haskell, The Carbohydrates vol. IIA, W. Pigman, D. Horton, Eds. (Academic Press, New York, 2nd ed., 1970) pp 159-172. Configuration: J. Daly et al., J. Am. Chem. Soc. 82, 5928 (1960); H. E. Carter et al., ibid. 83, 3723 (1961); R. U. Lemieux, R. J. Cushley, Can. J. Chem. 41, 858 (1963). Synthesis: M. Nakajima et al., Tetrahedron Lett. 1964, 967; T. Suami et al., ibid. 1967, 2671; S. Ogawa et al., J. Org. Chem. 39, 812 (1974); H. Prinzbach et al., Angew. Chem. Int. Ed. 14, 225 (1975); S. Ogawa et al., J. Org. Chem. 42, 3083 (1977). Review of syntheses: G. F. Busscher et al., Chem. Rev. 105, 775-791 (2005).