2932. 11-Desoxy-17-hydroxycorticosterone
Nomenclature
CAS number: 152-58-9
17,21-Dihydroxypregn-4-ene-3,20-dione; 17-hydroxydesoxycorticosterone; 4-pregnene-17α,21-diol-3,20-dione; 17-(1-keto-2-hydroxyethyl)-4-androsten-17α-ol-3-one; Reichstein's Substance S; 11-desoxycortisone; cortexolone. C21H30O4; mol wt 346.46.
C 72.80%, H 8.73%, O 18.47%.
Description and references
Isoln and partial synthesis: Reichstein, von Euw, Helv. Chim. Acta 21, 1197 (1938); Reichstein, ibid. 1490; Reichstein, von Euw, ibid. 23, 1258 (1940). Partial synthesis involving the chromic acid oxidation of 4-pregnene-17α,20,21-triol-3-one 21-monoacetate: Sarett, J. Biol. Chem. 162, 627 (1946). Prepn from 3α-formoxy-17α-hydroxypregnan-20-one: Gallagher et al., J. Am. Chem. Soc. 71, 3262 (1949); from 16,17-oxido-5-pregnen-3β-ol-20-one acetate: Julian et al., ibid. 3574; from 5-pregnen-3β-ol-20-one: Julian et al., ibid. 72, 5145 (1950).
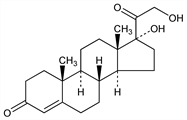
Properties
Fine, glistening plates from ether. mp 212.8-216.8°; gives no depression of melting point when mixed with cortisone. Very sparingly sol in water, ether. Sol in acetone, methanol, alcohol. Gives a carmine-red fluorescence reaction with concd. H2SO4. Reduces ammoniacal silver nitrate soln at room temp. uv max: 242 nm (E1%1cm 500). Oxidation with chromic acid in glacial acetic acid yields 4-androstene-3,17-dione.Derivative
Acetate.C23H32O5; mol wt 388.50.
C 71.11%, H 8.30%, O 20.59%.