2947. Dexpanthenol
Nomenclature
CAS number: 81-13-0
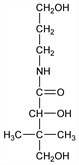
2,4-Dihydroxy-N-(3-hydroxypropyl)-3,3-dimethylbutanamide; d(+)-α,γ-dihydroxy-N-(3-hydroxypropyl)-β,β-dimethylbutyramide; pantothenylol; N-pantoyl-3-propanolamine; pantothenol; pantothenyl alcohol; Alcopan-250; Intrapan (USV); Pantenyl (Kay); Panthoderm (USV); Motilyn (Abbott); Bepanthen (Roche); Cozyme (Travenol); Ilopan (Warren-Teed); Urupan (Merckle). C9H19NO4; mol wt 205.25.
C 52.67%, H 9.33%, N 6.82%, O 31.18%.
Description and references
Prepd by the addition of propanolamine to optically active α,γ-dihydroxy-β,β-dimethylbutyrolactone: Schnider, Jubilee Vol. Emil Barell 1946, 85; CH 227706 (1943); GB 582156 (1946); US 2413077 (1946 to Hoffmann-La Roche). Only the d(+)-form has vitamin activity.
Properties
Viscous, somewhat hygroscopic liq. Slightly bitter taste. d2020 1.2. bp0.02 118-120°. Easily dec on distn. [α]D20 +29.5° (c = 5). nD20 1.497. Freely sol in water, alcohol, methanol. Slightly sol in ether. Natural pH about 9.5. Reasonably stable to usual sterilization time and temp in aq solns adjusted to pH 3.0-4.0, but long heating causes racemization. Hydrolyzed by alkali and strong acid. Usually more stable than salts of pantothenic acid if pH can be adjusted between 3 and 5. For add'l stability data see Rubin, J. Am. Pharm. Assoc. Sci. Ed. 37, 502 (1948). Aq solns can be stabilized with pantolactone: US 2898373 (1959).Derivative
dl-Form.Nomenclature
Panthenol.Therapeutic Category
Cholinergic; dl-form as vitamin.
Therapeutic Category (Veterinary)
Nutritional factor. Dietary source of pantothenic acid.Keywords
Cholinergic; Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B5