2955. Dextromoramide
Nomenclature
CAS number: 357-56-2
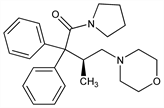
1-[(3S)-3-Methyl-4-(4-morpholinyl)-1-oxo-2,2-diphenylbutyl]pyrrolidine; (+)-1-(3-methyl-4-morpholino-2,2-diphenylbutyryl)pyrrolidine; 4-[2-methyl-4-oxo-3,3-diphenyl-4-(1-pyrrolidinyl)butyl]morpholine; d-2,2-diphenyl-3-methyl-4-morpholinobutyrylpyrrolidine; pyrrolamidol; R-875; SKF-5137. C25H32N2O2; mol wt 392.53.
C 76.50%, H 8.22%, N 7.14%, O 8.15%.
Description and references
Synthetic opioid analgesic. Synthesis: Janssen, J. Am. Chem. Soc. 78, 3862 (1956); GB 822055 (1959 to Janssen). Clinical pharmacokinetics: J. G. R. Ufkes et al., Pharm. World Sci. 20, 83 (1998). Clinical trial as adjuvant to methadone maintenance treatment: J. W. de Vos et al., Addict. Behav. 24, 707 (1999).
Properties
Crystals, mp 180-184°. [α]D20 +25.5° (c = 5 in benzene). uv max (0.01N isopropanol-HCl): 254, 260, 264 nm. Practically insol in water. Soly in 0.1N HCl: 1:25 (w/v). Soly (g/100 ml) in ethanol 50; in methanol 40; in acetone 50; in ethyl acetate 40; in benzene 5; in chloroform 5. Sol in ether.Derivative
Bitartrate.Nomenclature
CAS number: 2922-44-3
Palfium (Janssen). C25H32N2O2.C4H6O6; mol wt 542.62.
C 64.19%, H 7.06%, N 5.16%, O 23.59%.
Properties
Minute crystals, bitter taste. Dec 189-192°. Soly (w/v) at 25°: Water 20%, chloroform 30%, methanol 40%, ethanol 100%, acetone 100%.Note
This is a controlled substance (opiate): 21 CFR, 1308.11.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Diphenylpropylamine Derivatives