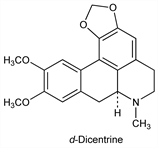
3041. Dicentrine
Nomenclature
CAS number: 517-66-8
6,7,7a,8-Tetrahydro-10,11-dimethoxy-7-methyl-5H-benzo[g]-1,3-benzodioxolo[6,5,4-de]quinoline; 9,10-dimethoxy-1,2-(methylenedioxy)aporphine; 1,2-methylenedioxy-9,10-dimethoxyaporphine. C20H21NO4; mol wt 339.39.
C 70.78%, H 6.24%, N 4.13%, O 18.86%.
Description and references
d-Form prevalent in nature. Found in Dicentra pusilla Sieb. & Zucc., Fumariaceae and several other Dicentra species. Related to actinodaphnine, and laurotetanine, q.q.v. Isoln: Y. Asahina, Arch. Pharm. 247, 201 (1909). Isoln of l-form from Duguetia A. St. Hil., Annonaceae: Casagrande, Ferrari, Farmaco Ed. Sci. 25, 442 (1970). Synthesis of dl-form: Haworth et al., J. Chem. Soc. 127, 2018 (1925); Cava et al., Tetrahedron 29, 2245 (1973). Resolution of isomers: Haworth et al., J. Chem. Soc. 129, 29 (1926).
Derivative
dl-Form.Properties
Prisms from methanol, mp 178-179°. Freely sol in chloroform, ethyl acetate, acetone, benzene, hot alcohol. Moderately sol in ether, cold alcohol. Absorption max: Girardet, J. Chem. Soc. 1931, 2630.Derivative
d-Form.Properties
Long prisms from ether. mp 169°. [α]D17 +64.1° (c = 1.433 in chloroform). Freely sol in alcohol, ethyl acetate, benzene.Derivative
l-Form.Properties
Long prisms from ether. mp 169°. [α]D17 -63.5° (c = 1.70 in chloroform).Derivative
dl-Hydrochloride.C20H21NO4.HCl; mol wt 375.85.
C 63.91%, H 5.90%, N 3.73%, O 17.03%, Cl 9.43%.
Properties
Small needles from water, dec 263-265°.Derivative
dl-Methiodide monohydrate.C20H21NO4.CH3I.H2O; mol wt 499.34.
C 50.51%, H 5.25%, N 2.81%, O 16.02%, I 25.41%.