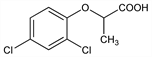
3078. Dichlorprop
Nomenclature
Description and references
Selective pre- and post-emergent herbicide. Exists in two optically active forms; only the (+)-isomer shows biological activity. First prepd and described as a plant growth regulator: M. E. Synerholm, P. W. Zimmerman, Contrib. Boyce Thompson Inst. 14, 91 (1945). Commercial prepn: H. A. Stevenson, R. F. Brookes, GB 822199 (1959 to Boots). Resoln of enantiomers and biological activity: S. T. Collins, F. E. Smith: J. Sci. Food Agric. 3, 248 (1952). Field studies and toxicology: W. O. G. Nuyken et al., Meded. Fac. Landbouwwet. Rijksuniv. Gent 52, 1139 (1987). Enantiomeric effects on degradation: A.W. Garrison et al., Environ. Sci. Technol. 30, 2449 (1996). GC/MS determn in soil: T. Heberer, H.-J. Stan, J. AOAC Int. 79, 1428 (1996).