3104. Dienestrol
Nomenclature
CAS number: 84-17-3
4,4′-(1,2-Diethylidene-1,2-ethanediyl)bisphenol; 4,4′-(diethylideneethylene)diphenol; 3,4-bis(p-hydroxyphenyl)-2,4-hexadiene; 4,4′-dihydroxy-γ,δ-diphenyl-β,δ-hexadiene; di(p-oxyphenyl)-2,4-hexadiene; dienoestrol; estrodienol; Cycladiene (Bruneau); Dienol; Dinovex (Ortho); DV (HMR); Estroral; Gynefollin; Hormofemin (Medo); Oestrasid; Oestrodiene; Oestroral; Restrol (Central Pharm.); Retalon; Synestrol (Schering). C18H18O2; mol wt 266.33.
C 81.17%, H 6.81%, O 12.01%.
Description and references
Synthesis: Dodds et al., Proc. Roy. Soc. 127B, 162 (1939); Hobday, Short, J. Chem. Soc. 1943, 609; Short, Hobday, US 2464203 (1949 to Boots); Adler, US 2465505 (1949 to Hoffmann-La Roche). Configuration: Koch, Nature 161, 309 (1948); Lane, Spialter, J. Am. Chem. Soc. 73, 4408 (1951).
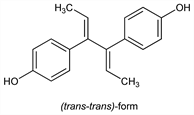
Properties
Minute needles from dil alcohol, mp 227-228°. Sublimes at 130° and 1 mm Hg. The sublimate mp 231-234°. Freely sol in alcohol, methanol, ether, acetone, propylene glycol; sol in chloroform, aq solns of alkali hydroxides; sol in vegetable oils after warming, but crystallizes out on standing. Practically insol in water, dil acids.Derivative
Diacetate.Nomenclature
CAS number: 84-19-5
Lipamone; Retalon-Oral. C22H22O4; mol wt 350.41.
C 75.41%, H 6.33%, O 18.26%.
Properties
Prisms from alc, mp 119-120°.Therapeutic Category
Estrogen.
Therapeutic Category (Veterinary)
Estrogenic hormone therapy.Keywords
Estrogen; Nonsteroidal