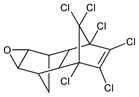
3103. Dieldrin
Nomenclature
CAS number: 60-57-1
(1aα,2β,2aα,3β,6β,6aα,7β,7aα)-3,4,5,6,9,9-Hexachloro-1a,2,2a,3,6,6a,7,7a-octahydro-2,7:3,6-dimethanonaphth[2,3-b]oxirene; 1,2,3,4,10,10-hexachloro-6,7-epoxy-1,4,4a,5,6,7,8,8a-octahydro-endo,exo-1,4:5,8-dimethanonaphthalene; insecticide no. 497; HEOD; ENT-16225; compd 497; Octalox (Velsicol). C12H8Cl6O; mol wt 380.91.
C 37.84%, H 2.12%, Cl 55.84%, O 4.20%.
Description and references
Stereoisomer of endrin, q.v. Activity: C. W. Kearns et al., J. Econ. Entomol. 42, 127 (1949). Prepn from aldrin, q.v.: Soloway, US 2676131 (1954 to Shell); Payne, Smith, US 2776301 (1957 to Shell). Synthesis: Korte, Rechmeier, Ann. 656, 131 (1962). Metabolism: M. K. Baldwin et al., Chem. Ind. (London) 1970, 595; C. T. Bedford, D. H. Hutson, ibid. 1976, 440. Toxicity data: Gaines, Toxicol. Appl. Pharmacol. 14, 515 (1969). Review of toxicology and human exposure: Toxicological Profile for Aldrin/Dieldrin (PB2003-100134, 2002) 354 pp.