3108. Diethazine
Nomenclature
CAS number: 60-91-3
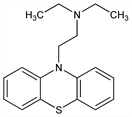
N,N-Diethyl-10H-phenothiazine-10-ethanamine; 10-(2-diethylaminoethyl)phenothiazine; N-(diethylaminoethyl)thiodiphenylamine; N-(2′-diethylaminoethyl)dibenzoparathiazine; RP-2987; Deparkin; Dinezin; Dolisina; Eazaminum; Ethylemin; Parkazin. C18H22N2S; mol wt 298.45.
C 72.44%, H 7.43%, N 9.39%, S 10.74%.
Description and references
Anticholinergic. Prepd by reacting 10-phenothiazineethyl chloride with diethylamine in presence of copper powder or by reacting diethylaminoethyl chloride with phenothiazine: Charpentier, Compt. Rend. 225, 306 (1947); Huttrer, Enzymologia 12, 293 (1948); Charpentier, US 2530451 (1950 to Rh^one-Poulenc); Berg, Ashley, US 2607773 (1952 to Rh^one-Poulenc). Toxicity study: Bovet et al., Therapie 2, 115 (1947).
Properties
Oily liquid. bp4-5 195-208°; bp0.4-0.5 167-175°.Derivative
Hydrochloride.Nomenclature
CAS number: 341-70-8
Antipar (Farmitalia); Aparkazin; Diparcol (Specia); Latibon (Bayer); Thiantan; Thiontan. C18H22N2S.HCl; mol wt 334.91.
C 64.55%, H 6.92%, N 8.36%, S 9.57%, Cl 10.59%.
Properties
Crystals, mp 184-186°. Burning taste, producing a temporary numbness of the tongue. One part dissolves in about 5 parts water, 6 parts ethanol, 5 parts chloroform. Practically insol in ether. pH of 10% aq soln 5.0-5.3. LD50 orally in mice: 450 mg/kg (Bovet).Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian; Antimuscarinic