3116. Diethylcarbamazine
Nomenclature
CAS number: 90-89-1
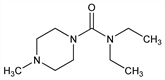
N,N-Diethyl-4-methyl-1-piperazinecarboxamide; 1-diethylcarbamoyl-4-methylpiperazine; carbamazine; 1-diethylcarbamyl-4-methylpiperazine; 84L; RP-3799; Carbilazine; Caricide (Am. Cyanamid); Cypip (Am. Cyanamid); Ethodryl (BDH); Notézine (Specia); Spatonin. C10H21N3O; mol wt 199.29.
C 60.27%, H 10.62%, N 21.08%, O 8.03%.
Description and references
Prepn: Kushner et al., J. Org. Chem. 13, 151 (1948); Kushner, Brancone, US 2467893; US 2467895 (1949 to Am. Cyanamid). Pharmacology and toxicology: Harned et al., Ann. N.Y. Acad. Sci. 50, 141 (1948). GC determn in blood: S. Nene et al., J. Chromatogr. 308, 334 (1984). Mode of action: J.-Y. Cesbron et al., Nature 325, 533 (1987). Review of pharmacology, mechanisms of action and clinical uses: C. D. MacKenzie, M. A. Kron, Trop. Dis. Bull. 82(10), R1-R37 (1985).
Properties
Crystals, mp 47-49°. bp3 108.5-111°.Derivative
Hydrochloride.C10H21N3O.HCl; mol wt 235.75.
C 50.95%, H 9.41%, N 17.82%, O 6.79%, Cl 15.04%.
Properties
Crystals from acetone, mp 156.5-157°. Very sol in water; sol in chloroform, dioxane.Derivative
Citrate.Nomenclature
CAS number: 1642-54-2
Banocide (Wellcome); Dec (Wendt); Dirocide (Solvay); Filaribits (SKB); Filazine; Franocide (Burroughs Wellcome); Hetrazan (Lederle); Loxuran (EGYT); Longicid (Chassot). C10H21N3O.C6H8O7; mol wt 391.42.
C 49.10%, H 7.47%, N 10.74%, O 32.70%.
Properties
Crystals, mp 141-143°. Freely sol in water (>75% at 20°). Sparingly sol in cold alc; freely sol in hot alc. Practically insol in benzene, acetone, ether, chloroform. LD50 orally in rats: 1.38 g/kg (Harned).Derivative
Phosphate.Nomenclature
Ditrazin.Properties
Crystals, freely sol in water.Therapeutic Category
Anthelmintic (Nematodes).
Therapeutic Category (Veterinary)
Citrate as an anthelmintic.Keywords
Anthelmintic (Nematodes)