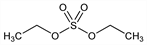
3130. Diethyl Sulfate
Nomenclature
CAS number: 64-67-5
Sulfuric acid diethyl ester. C4H10O4S; mol wt 154.18.
C 31.16%, H 6.54%, O 41.51%, S 20.80%.
Description and references
Prepd from ethanol + sulfuric acid; by absorption of ethylene in sulfuric acid; from diethyl ether and fuming sulfuric acid. Review of prepn and uses: R. Page, J. A. John in Ethylene and its Industrial Derivatives S. A. Miller, Ed. (Benn, London, 1969) pp 774-787. Toxicity data: H. F. Smyth et al., J. Ind. Hyg. Toxicol. 31, 60 (1949). Review of carcinogenicity studies: IARC Monographs 4, 277-281 (1974). Reviews: C. M. Suter, The Organic Chemistry of Sulfur (John Wiley, 1944) pp 62-65; E. E. Gilbert, Sulfonation and Related Reactions (Interscience, New York, 1965).