3196. Diisopropylamine
Nomenclature
CAS number: 108-18-9
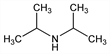
N-(1-Methylethyl)-2-propanamine. C6H15N; mol wt 101.19.
C 71.22%, H 14.94%, N 13.84%.
Description and references
Prepn: A. Siersch, Ann. 148, 263 (1868); M. Van der Zande, Rec. Trav. Chim. 8, 202 (1889). Enthalpy of formation: M. A. V. Ribeiro da Silva et al., J. Chem. Thermodyn. 29, 1025 (1997). Toxicity study: H. F. Smyth et al., Arch. Ind. Hyg. Occup. Med. 10, 61 (1954). Safety assessment: J. Am. Coll. Toxicol. 14, 182-192 (1995).
Properties
Liquid; characteristic odor; strongly alkaline. d22 0.722. bp 84°. Flash pt, open cup: 21°F (-6°C). Sol in water, alc. LD50 orally in rats: 0.77 g/kg (Smyth).Derivative
Lithium salt.Nomenclature
CAS number: 4111-54-0
LDA; lithiodiisopropylamine; lithium diisopropylamide. Description and references
Strong nucleophile. Crystal structure: N. D. R. Barnett et al. J. Am. Chem. Soc. 113, 8187 (1991). Use as nucleophile: Y. Tanaka et al., Bull. Chem. Soc. Jpn. 60, 788 (1987); S. Saito et al., J. Am. Chem. Soc. 119, 611 (1997). Review of chemistry: Reagents for Organic Synthesis vol. 8, M. Fieser, Ed. (Wiley-Interscience, New York, 1980) pp 292-299.