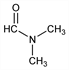
3243. N,N-Dimethylformamide
Nomenclature
Description and references
Prepd from dimethylamine and formic acid: Mitchell, Reid, J. Am. Chem. Soc. 53, 1879 (1931); Brown, J. Appl. Chem. 1, Suppl. Issue no. 2, S159 (1951); Campbell, US 3015674 (1962 to Commercial Solvents); Surman, US 3072725 (1963 to du Pont); from dimethylamine + HCN: Benneville et al., J. Org. Chem. 21, 772 (1956); from HCN + methanol: Fukuoka, Kominami, Chem. Tech. 1972 (Nov.), 640. Toxicity study: W. Bartsch et al., Arzneim.-Forsch. 26, 1581 (1976). Reviews of chemical uses: R. S. Kittila, Dimethylformamide Chemical Uses (du Pont, Wilmington, 1967) 264 pp and Suppl. (1973) 148 pp; J. S. Pizey, Synthetic Reagents Vol. 1 (John Wiley, New York, 1974) pp 4-99; C. L. Eberling in Kirk-Othmer Encyclopedia of Chemical Technology vol. 11 (Wiley-Interscience, New York, 3rd ed., 1980) pp 263-268. Review of carcinogenic risk: IARC Monographs 47, 171-197 (1989).