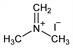3251. Dimethyl(methylene)ammonium Iodide
Nomenclature
CAS number: 33797-51-2
N-Methyl-N-methylenemethanaminium iodide; Eschenmoser's salt. C3H8IN; mol wt 185.01.
C 19.48%, H 4.36%, I 68.59%, N 7.57%.
Description and references
Mannich type intermediate originally developed to introduce methyl groups into corrin chromophore. Prepn from trimethylamine and diiodomethane: J. Schreiber et al., Angew. Chem. Int. Ed. 10, 330 (1971); from N,N,N′,N′-tetramethylmethylenediamine: T. A. Bryson et al., J. Org. Chem. 45, 524 (1980). Used in prepn of Mannich bases: J. Hooz, J. N. Bridson, J. Am. Chem. Soc. 95, 602 (1973). In functionalization of indoles: A. P. Kozikowski, H. Isida, Heterocycles 14, 55 (1980). In prepn of α-methylene carbonyls: J. L. Roberts et al., Tetrahedron Lett. 1977, 1621; of terminal olefins: eidem, ibid. 1299.