3287. Dinoseb
Nomenclature
CAS number: 88-85-7
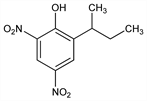
2-(1-Methylpropyl)-4,6-dinitrophenol; 2-sec-butyl-4,6-dinitrophenol; DNBP; ENT-1122; WSX-8365; Chemox PE (Blue Spruce); Dow General (Dow); Premerge (Dow); Subitex (Hoechst); Caldon (Hoechst); Basanite (BASF). C10H12N2O5; mol wt 240.21.
C 50.00%, H 5.04%, N 11.66%, O 33.30%.
Description and references
Prepn: L. E. Mills, B. L. Fayerweather, US 2192197 (1940 to Dow). Activity: A. S. Crafts, Science 101, 417 (1945). Metabolism: S. K. Bandal, J. E. Casida, J. Agric. Food Chem. 20, 1235 (1972); K. Ingebrigtsen, A. Froeslie, Acta Pharmacol. Toxicol. 46, 326 (1980). Toxicology: R. G. Bough et al., Toxicol. Appl. Pharmacol. 7, 353 (1965); T. B. Gaines, R. E. Linder, Fundam. Appl. Toxicol. 7, 299 (1986).
Properties
Orange-brown viscous liquid, mp 38-42°. pKa 4.62. Sol in most organic solvents. LD50 in adult male, female rats (mg/kg): 27, 28 orally (Gaines, Linder).Derivative
Acetate.Nomenclature
CAS number: 2813-95-8
HOE-2904; Aretit (Hoechst); Ivosit (Hoechst). C12H14N2O6; mol wt 282.25.
C 51.06%, H 5.00%, N 9.93%, O 34.01%.
Properties
Brown oil, vinegar-like odor, mp 26-27°. Vapor press at 20°: 6 × 10-4 mm Hg. Sol in aromatics.Derivative
Ammonium salt.Nomenclature
CAS number: 6365-83-9
Chemox Selective (Blue Spruce); Dow Selective (Dow); Sinox W (FMC). C10H15N3O5; mol wt 257.24.
C 46.69%, H 5.88%, N 16.33%, O 31.10%.
Derivative
Triethanolamine salt.Nomenclature
CAS number: 6420-47-9
DN-289; Chemox DN (Blue Spruce); Gebutox (Hoechst). C16H27N3O8; mol wt 389.40.
C 49.35%, H 6.99%, N 10.79%, O 32.87%.