3337. Dipipanone
Nomenclature
CAS number: 467-83-4
4,4-Diphenyl-6-(1-piperidinyl)-3-heptanone; dl-4,4-diphenyl-6-piperidinoheptan-3-one; 6-piperidino-4,4-diphenylheptan-3-one; 2-(1-piperidino)-4,4-diphenyl-5-heptanone; phenylpiperone; piperidylamidone. C24H31NO; mol wt 349.51.
C 82.47%, H 8.94%, N 4.01%, O 4.58%.
Description and references
Opioid analgesic. Prepn: Ofner, Walton, J. Chem. Soc. 1950, 2158; GB 654975 (1951 to Wellcome Foundation). Variations of the synthesis using phenyllithium as condensing agent: Kazuhiko, Kubota, Kumamoto Med. J. 12, 304-307 (1960). GC determn in plasma, urine: S. Paterson, J. Chromatogr. B 424, 152 (1988).
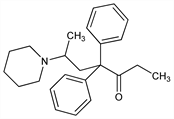
Derivative
Hydrochloride.Nomenclature
CAS number: 75783-06-1
Hoechst 10805. C24H31NO.HCl; mol wt 385.97.
C 74.68%, H 8.36%, N 3.63%, O 4.15%, Cl 9.19%.
Properties
Component of Diconal. Minute prisms from wet alcohol-ether, mp 123-126°. Also reported as mp 126-127°.Derivative
Hydrobromide.Nomenclature
CAS number: 909260-86-2
C24H31NO.HBr; mol wt 430.42.
C 66.97%, H 7.49%, N 3.25%, O 3.72%, Br 18.56%.
Properties
Crystals from water, mp 103-106°.Note
This is a controlled substance (opiate): 21 CFR, 1308.11.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Diphenylpropylamine Derivatives