3397. Docetaxel
Nomenclature
Description and references
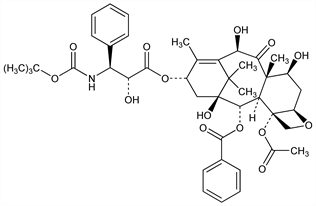
Semisynthetic derivative of paclitaxel, q.v., prepd using a natural precursor, 10-deacetylbaccatin III, extracted from the needles of the European yew tree, Taxus baccata L., Taxaceae. Antimitotic agent that promotes the assembly of microtubules and inhibits their depolymerization to free tubulin. Prepn: M. Colin et al., EP 253738; eidem, US 4814470 (1988, 1989 both to Rh^one-Poulenc Sante); L. Mangatal et al., Tetrahedron 45, 4177 (1989). Synthesis of the side chain: J.-N. Denis et al., J. Org. Chem. 56, 6939 (1991). Structure-activity study: F. Guéritte-Voegelein et al., J. Med. Chem. 34, 992 (1991). HPLC determn and degradation studies in pharmaceutical formulations: D. Kumar et al., J. Pharm. Biomed. Anal. 43, 1228 (2007). LC-MS determn in plasma: R. A. Parise et al., J. Chromatogr. B 783, 231 (2003). Review of clinical pharmacokinetics, metabolism, and analytical methods: S. D. Baker et al., Clin. Pharmacokinet. 45, 235-252 (2006). Review of mechanism of action and synergistic therapies: R. S. Herbst, F. R. Khuri, Cancer Treat. Rev. 29, 407-415 (2003); of clinical experience with solid tumors: A. Montero et al., Lancet Oncol. 6, 229-239 (2003). Review of alternative formulations: F. K. Engels et al., Anti-Cancer Drugs 18, 95-103 (2007).
Properties
mp 232°. [α]D -36° (c = 0.74 in ethanol). uv max: 230, 275, 283 nm (ε 14800, 1730, 1670).Derivative
Trihydrate.Nomenclature
Properties
White to almost-white powder. Highly lipophilic. Practically insol in water.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkaloids/Natural Products; Taxanes