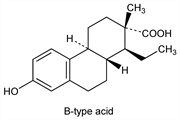
3410. Doisynolic Acid
Nomenclature
Description and references
Prepd by alkali fusion of estrone: MacCorquodale et al., J. Biol. Chem. 99, 327 (1933); 101, 753 (1933); Heer, Miescher, Helv. Chim. Acta 28, 156 (1945). Sixteen isomers are possible. If the hydrogen atom at position 4a is α- and the hydrogen atom at position 10a is β-, the acid belongs to the A series. If the hydrogen at 4a is β- and the hydrogen at 10a is α-, the acid belongs to the B series. If both hydrogens are α-, the acid belongs to the C series. If both hydrogens are β-, the acid belongs to the D series: Miescher, Chem. Rev. 43, 367 (1948). If the hydrogen atom at position 1 and the methyl group at position 2 have either the α,α- or β,β-orientation, the acid is designated cis; if they are α,β- the acid is designated trans: L. F. Fieser, M. Fieser, Steroids (Reinhold, New York, 1959) pp 487-495. Synthesis of dl-cis and dl-trans of the A series and dl-cis of the B series: Anner, Miescher, Helv. Chim. Acta 29, 1889 (1946); 30, 1422 (1947). Synthesis of dl-cis of the C series: Jilck, Protiva, Collect. Czech. Chem. Commun. 23, 692 (1958). Stereochemistry of d-cis-form: Iriarte, Crabbe, Chem. Commun. 1972, 1110. Reviews: Shoppee, Annu. Rep. Prog. Chem. 44, 190 (1948); Miescher in Recent Progress in Hormone Research vol. 3, G. Pincus Ed. (Academic Press, New York, 1948) pp 47-69.