3411. Dolasetron
Nomenclature
Description and references
Bridged pseudopelletierine derivative; specific serotonin (5HT3) receptor antagonist. Prepn: M. W. Gittos, EP 266730; idem et al., US 4906755 (1988, 1990 both to Merrell Dow). Binding study: P. H. Boeijinga et al., Eur. J. Pharmacol. 219, 9 (1992). Pharmacology: R. C. Miller et al., Drug Dev. Res. 28, 87 (1993). Clinical pharmacokinetics: H. Boxenbaum et al., Biopharm. Drug Dispos. 13, 693 (1992); eidem, ibid. 14, 131 (1993). GC-MS and LC determn in plasma: T. A. Gillespie et al., J. Pharm. Biomed. Anal. 11, 955 (1993). Clinical trial in emetogenic chemotherapy: A. A. Fauser et al., Eur. J. Cancer 32A, 1523 (1996); for prevention of postoperative nausea: S. G. Graczyk et al., Anesth. Analg. 84, 325 (1997); B. K. Philip et al., J. Clin. Anesth. 12, 1 (2000). Review of veterinary use: G. K. Ogilvie, J. Am. Anim. Hosp. Assoc. 36, 481-483 (2000).
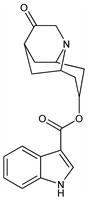
Derivative
Methanesulfonate.Nomenclature
Properties
Crystalline solid, mp 278°. Freely sol in water, propylene glycol; slightly sol in alcohol.Therapeutic Category
Antiemetic.
Therapeutic Category (Veterinary)
Antiemetic.Keywords
Antiemetic; Serotonin Receptor Antagonist