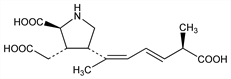
3417. Domoic Acid
Nomenclature
Description and references
Excitatory amino acid isolated from the red alga Chondria armata Okamura, Rhodomelaceae, known in Japanese as “domoi”. Structural analog of kainic acid, q.v. Shown to be responsible for amnesic shellfish poisoning associated with ingestion of certain cultured blue mussels. Isoln: T. Takemoto, K. Daigo, Chem. Pharm. Bull. 6, 578 (1958); eidem, Arch. Pharm. 293, 627 (1960). Series of articles on isoln, structure, and anthelminthic activity: K. Daigo, Yakugaku Zasshi 79, 350-364 (1959), C.A. 53, 14218 (1959). Structural studies: T. Takemoto et al., ibid. 86, 874 (1966), C.A. 66, 28604m (1967). Total synthesis and revised structure: Y. Ohfune, M. Tomita, J. Am. Chem. Soc. 104, 3511 (1982). Neuroexcitatory activity: T. J. Biscoe et al., Nature 255, 166 (1975); R. Zaczek, J. T. Coyle, Neuropharmacology 21, 15 (1982). Effect on kainate receptor activation: G. Debonnel et al., Can. J. Physiol. Pharmacol. 67, 29, 904 (1989). Identification in toxic mussel extracts: J. L. C. Wright et al., Can. J. Chem. 67, 481 (1989). LC determn in shellfish products: J. F. Lawrence et al., J. Chromatogr. 462, 349, 419 (1989). Reviews: M. A. Quilliam, J. L. C. Wright, Anal. Chem. 61, 1053A-1059A (1989); J. Clayden et al., Tetrahedron 61, 5713-5724 (2005).