5276. Kainic Acid
Nomenclature
Description and references
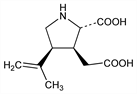
Anthelmintic principle from the dried red alga Digenea simplex (Wulf.) Ag., Rhodomelaceae: Murakami et al., J. Pharm. Soc. Jpn. 73, 1026 (1953); JP 54 4947 (1954), C.A. 49, 13604i (1955); Katsuya et al., JP 64 1942 (1964 to New-Japan Pharmaceutical Co). Structure: Watase, Nitta, Bull. Chem. Soc. Jpn. 30, 889 (1957); Watase et al., ibid. 31, 714 (1958). Eight theoretical stereoisomers, Nitta et al., Nature 181, 761 (1958); GB 795750 (1958 to Takeda). Synthesis: Ueno, US 2902492; Tatsuoka et al., US 2954384 (1959, 1960 both to Takeda); W. Oppolzer, H. Andres, Helv. Chim. Acta 62, 2282 (1979). Excitotoxic amino acid used to identify a specific subset of EAA receptors. Consequently the receptors are known as kainate receptors. Neurotoxic activity: J. V. Nadler, Life Sci. 24, 289 (1979). Mechanism of neurotoxicity: E. G. McGeer et al., Adv. Neurol. 23, 577 (1979); J. T. Coyle et al., ibid. 593; J. W. Ferkany et al., Nature 298, 757 (1982); J. Garthwaite, G. Garthwaite, ibid. 305, 138 (1983). Induces epileptogenic lesions: J. V. Nadler, Neurosci. Res. Program Bull. 19, 369 (1981). Autoradiographic characterization of binding sites: J. T. Greenamyre et al., J. Pharmacol. Exp. Ther. 233, 254 (1985). Book: Kainic Acid as a Tool in Neurobiology, E. G. McGeer et al., Eds. (Raven Press, New York, 1978). Review: J. T. Coyle, Ciba Found. Symp. 126, 186-203 (1987).
Properties
Needles, dec 251°. [α]D24 14.8° (c = 1.01). Intense absorption at 6.05 and 11.2 μ. Sol in water. Insol in ethanol. Stable in boiling aq solns.Use
Neurobiological tool.Therapeutic Category
Anthelmintic (Nematodes).
Keywords
Anthelmintic (Nematodes)