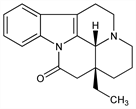
3487. Eburnamonine
Nomenclature
Description and references
One of the Vinca alkaloids, naturally occurring as the (+)- and (±)-forms. Isoln of the (+)-form from Hunteria eburnea Pinchon, Apocynaceae: M. F. Bartlett et al., Compt. Rend. 249, 1259 (1959); of the (±)-form from Vinca minor L., Apocynaceae: J. Mokry et al., Experientia 17, 354 (1961). The (-)-form is obtained by acid hydrolysis of vincamine, q.v.: J. Trajanek et al., Tetrahedron Lett. 1961, 702; O. Clauder et al., ibid. 1962, 1147; eidem, HU 151295 (1964 to Gedeon Richter), C.A. 60, 14558e (1964). Structure and synthesis of the (±)-form: M. F. Bartlett, W. I. Taylor, Tetrahedron Lett. 20, 20 (1959); eidem, J. Am. Chem. Soc. 82, 5941 (1960); E. Wenkert, B. Wickberg, ibid. 87, 1580 (1965). Short synthesis of the (±)-form: E. Wenkert et al., ibid. 100, 4893 (1978); high yield total synthesis: J. L. Hermann et al., ibid. 101, 1540 (1979); T. Imanishi et al., Chem. Pharm. Bull. 30, 1521 (1982). Synthesis of the (-)-form: D. Cartier et al., Bull. Soc. Chim. Fr. 1976, 1961. Structural and biogenetic relationship to vincamine: J. Mokry et al., Tetrahedron Lett. 1962, 433. Pharmacology of the (-)-form: P. Lacroix et al., Arzneim.-Forsch. 29, 1094 (1979). Series of articles on the pharmacodynamics, metabolism, and therapeutic use of the (-)-form: Eur. Neurol. 17, Suppl. 1, 1-172 (1978). Reviews: W. I. Taylor in The Alkaloids vol. 8, R. H. F. Manske, Ed. (Academic Press, NewYork, 1965) pp 253-259; idem, ibid. vol. 11, pp 108-110 (1968).
Properties
Cryst, mp 174°. [α]D25 +89° (chloroform). uv max: 241, 268, 296, 302 nm (ε 19800, 10200, 4800, 4800).Derivative
(±)-Form.Nomenclature
Properties
Crystals from methanol, mp 201-202.5°. uv max (ethanol): 227, 287, 294 nm (log ε 4.49, 3.89, 3.87).Derivative
(-)-Form.Nomenclature
Properties
Solid, mp 168-170°; also reported as mp 177-178° (Cartier). [α]D25 -102° (chloroform) (Clauder); [α]D -100° (c = 0.783 in chloroform) (Cartier). uv max: 205, 240, 265, 290, 300 nm (log ε 4.28, 4.16, 3.90, 3.59, 3.57).Derivative
(-)-Form phosphate.Nomenclature
Therapeutic Category
The (-)-form as vasodilator.
Keywords
Vasodilator (Cerebral)