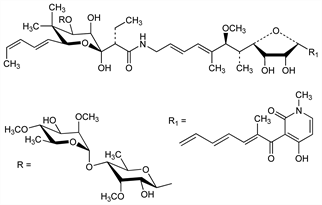
3525. Efrotomycin
Nomenclature
Description and references
Antibiotic produced by Streptomyces lactamdurans NRRL 3802: R. G. Wax, W. M. Maiese, DE 2450813 (1975 to Merck & Co.), C.A. 83, 145755y (1975); R. G. Wax et al., J. Antibiot. 29, 670 (1976). In vitro and in vivo activity: B. M. Frost et al., ibid. 1083; 32, 626 (1979). Production and growth promoting activity: W. M. Maiese, R. G. Wax, US 4024251 (1977 to Merck & Co.). Synergism with bottromycin, q.v.: B. M. Frost et al., J. Antibiot. 32, 1046 (1979). Structure: R. S. Dewey et al., ibid. 38, 1691 (1985). Stereospecific total synthesis: R. E. Dolle, K. C. Nicolaou, J. Am. Chem. Soc. 107, 1691, 1695 (1985). HPLC determn in feeds: J. D. Strong, Analyst 111, 853 (1986). Effect on gain and feed efficiency in swine: A. G. Foster et al., J. Anim. Sci. 65, 877 (1987).