3524. Efonidipine
Nomenclature
CAS number: 111011-63-3
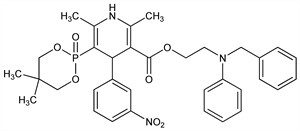
5-(5,5-Dimethyl-1,3,2-dioxaphosphorinan-2-yl)-1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-3-pyridinecarboxylic
acid 2-[phenyl(phenylmethyl)amino]ethyl ester, P-oxide; 2-(N-benzylanilino)ethyl (±)-1,4-dihydro-2,6-dimethyl-4-(m-nitrophenyl)-5-phosphononicotinate, cyclic 2,2-dimethyltrimethylene
ester. C34H38N3O7P; mol wt 631.66.
C 64.65%, H 6.06%, N 6.65%, O 17.73%, P 4.90%.
Description and references
Dihydropyridine calcium channel blocker. Prepn: K. Seto et al., WO 8704439; idem et al., US 4885284 (1987, 1989 both to Nissan); and crystal structure: R. Sakoda et al., Chem. Pharm. Bull. 40, 2362 (1992). Stereoselective synthesis of enantiomers and crystal structure of (S)-form: idem et al., ibid. 2377. Pharmacology: C. Shudo et al., J. Pharm. Pharmacol. 45, 525 (1993). Mechanism of action study: T. Yamashita et al., Jpn. J. Pharmacol. 57, 337 (1991). Clinical study: T. Saito et al., Curr. Ther. Res. 52, 113 (1992).
Properties
Crystals from ethyl acetate, mp 169-170° (Sakoda); also reported as mp 155-156° (Seto).Derivative
Hydrochloride.Nomenclature
CAS number: 111011-53-1
C34H38N3O7P.HCl; mol wt 668.12.
C 61.12%, H 5.88%, N 6.29%, O 16.76%, P 4.64%, Cl 5.31%.
Properties
LD50 in mice (mg/kg): >600 orally (Seto).Derivative
Hydrochloride ethanol.Nomenclature
CAS number: 111011-76-8
NZ-105; Landel (Zeria). C34H38N3O7P.C2H5OH.HCl; mol wt 714.18.
C 60.54%, H 6.35%, N 5.88%, O 17.92%, P 4.34%, Cl 4.96%.
Properties
Yellow crystals from aq ethanol, mp 151° (dec).Derivative
(S)- or (R)-Form.Properties
Pale yellow crystals from ethanol, mp 190-192°. [α]D25 + or -7.0° resp (c = 0.50 in chloroform).Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive; Dihydropyridine Derivatives; Calcium Channel Blocker; Dihydropyridine Derivatives