3547. Ellagic Acid
Nomenclature
Description and references
Occurs free or combined in galls. Isoln from the kino of Eucalyptus maculata Hook and E. hemipholia F. Muell., Myrtaceae: Gell et al., Aust. J. Chem. 11, 372 (1958); Hills, Carle, ibid. 16, 147 (1963). Prepd by sodium persulfate oxidation of gallic acid or by acid hydrolysis of crude tannin from walnuts: Perkin, Nierenstein, J. Chem. Soc. 87, 1412 (1905); Jurd, J. Am. Chem. Soc. 78, 3445 (1956); 79, 6043 (1957). Purification: FR 1478523 (1967 to Prod. Chim. Celluloses Rey), C.A. 68, 78267r (1968). Physicochemical properties: Press, Hardcastle, J. Appl. Chem. 19, 247 (1969). Pharmacology: Bhargava et al., J. Pharm. Sci. 57, 1728 (1968). Ellagic acid is a potent antagonist of the mutagenicity of bay-region diol epoxides of several aromatic hydrocarbons. Inhibition of the mutagenicity of the ultimate carcinogenic metabolite of benzo[a]pyrene: A. W. Wood et al., Proc. Natl. Acad. Sci. USA 79, 5513 (1982). Study of the reaction between this metabolite and ellagic acid: J. M. Sayer et al., J. Am. Chem. Soc. 104, 5562 (1982). Brief discussion of ellagic acid as a prototype of a new class of cancer-preventing drugs: J. Fox, Chem. Eng. News 60, 26 (Oct. 25, 1982).
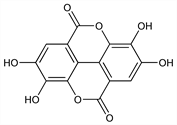
Properties
Cream colored needles from pyridine, mp >360°. uv max (ethanol): 366, 255 nm (log ε 3.93, 4.60). Slightly sol in water or alcohol; sol in alkalies, in pyridine. Practically insol in ether.Derivative
Tetraacetate.Properties
Needles from acetic anhydride, mp 340°.Therapeutic Category
Hemostatic.
Keywords
Hemostatic