3570. Encainide
Nomenclature
Description and references
Anti-arrhythmic benzanilide derivative. Prepn: S. J. Dykstra, J. L. Minielli, DE 2210154 (1972 to Bristol-Myers), C.A. 78, 4138j (1973); eidem, US 3931195 (1976 to Mead Johnson); S. J. Dykstra et al., J. Med. Chem. 16, 1015 (1973). Anti-arrhythmic pharmacology in animals: J. E. Byrne et al., J. Pharmacol. Exp. Ther. 200, 147 (1977). Clinical pharmacology and efficacy in chronic ventricular arrhythmia: D. M. Roden et al., N. Engl. J. Med. 302, 877 (1980). Electrophysiology and effects on cardiac conduction: M. Sami et al., Am. J. Cardiol. 44, 526 (1979). Hemodynamic effects: M. Sami et al., ibid. 52, 507 (1983). Adverse effects: R. A. Winkle et al., Am. Heart J. 102, 857 (1981). Comparison with other Class I anti-arrhythmic agents: A. Pottage, Am. J. Cardiol. 52, 24C (1983). Series of articles on pharmacology, pharmacokinetics, metabolism, clinical safety and efficacy: ibid. 58(5), 1C-116C (1986). Review: L. B. Mitchell, R. A. Winkle in New Drugs Annual: Cardiovascular Drugs vol. 1, A. Scriabine, Ed. (Raven Press, New York, 1983) pp 93-107.
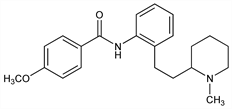
Derivative
Hydrochloride.Nomenclature
Properties
Crystals, mp 131.5-132.5°. Freely sol in water; slightly sol in ethanol. Insol in heptane. LD50 in mice, dogs (mg/kg): 86, 43 orally; 16, 17 i.v. (Mitchell, Winkle).Therapeutic Category
Antiarrhythmic (class IC).
Keywords
Antiarrhythmic