Description and references
A group of novel polycyclic compounds isolated
from leaves of the ‘Dorrigo Plum’, Endiandra introrsa C.
T. White, Lauraceae, a large tree occurring in rain forests
of Australia. Although they have a number of asymmetric centers,
endiandric acids occur in nature in racemic rather than enantiomeric
forms. This fact led to a proposed hypothesis for the “biogenesis”
of these compounds in nature from achiral precursors by a series of
non-enzymatic electrocyclizations called the “endiandric acid cascade”.
Isoln from E. introrsa leaves (obtained in 1958) and structure
of endiandric acid A: W. M. Bandaranayake et al., Chem. Commun. 1980, 162. Postulated
electrocyclic reactions, prediction of endiandric acid D as a natural
product: eidem, ibid. 902. X-ray crystallographic
structural elucidation of A and detailed report of isoln: eidem, Aust. J. Chem. 34, 1655 (1981). Isoln and structure of endiandric acid B: eidem, ibid. 35, 557 (1982);
of C: eidem, ibid. 567. Stereocontrolled total
synthesis of A and B, description of “endiandric acid cascade”: K.
C. Nicolaou et al., J. Am. Chem.
Soc. 104, 5555 (1982). Stereocontrolled total
synthesis of endiandric acids C-G: eidem, ibid. 5557. Synthesis of precursors and thermal studies: eidem, ibid. 5558, 5560.
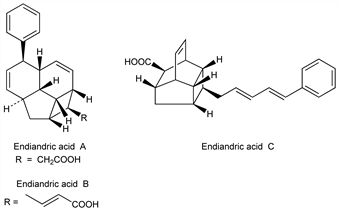
Derivative
Endiandric Acid A.
Nomenclature
CAS number: 74591-03-0
(1
R,1a
R,2a
R,5
S,5a
S,7a
S,7b
R,7c
R)-
rel-1a,2,2a,5,5a,7a,7b,7c-Octahydro-5-phenyl-1
H-cyclobut[
bc]acenaphthylene-1-acetic acid; 2-(6′-phenyltetracyclo[5,4,2,0
3,13,0
10,12]trideca-4′,8′-dien-11′-yl)acetic acid.
C
21H
22O
2; mol wt 306.40.
C 82.32%, H 7.24%, O 10.44%.
Properties
Rods from aq ethanol, mp 147-149°. uv max (95%
ethanol): 242, 255, 261, 268, 286 nm (log ε 2.19, 2.36, 2.45, 2.32, 1.45). pKa 5.1, 5.0. Gives a yellow color with tetranitromethane.Derivative
Endiandric Acid B.
Nomenclature
CAS number: 76060-33-8
(2
E)-
rel-4-[(1
R,1a
R,2a
R,5
S,5a
S,7a
S,7b
R,7c
R)-1a,2,2a,5,5a,7a,7b,7c-Octahydro-5-phenyl-1
H-cyclobut[
bc]acenaphthylen-1-yl]-2-butenoic acid; (
E)-4-(6′-phenyltetracyclo[5,4,2,0
3,13,0
10,12]trideca-4′,8′-dien-11′-yl)but-2-enoic acid.
C
23H
24O
2; mol wt 332.44.
C 83.10%, H 7.28%, O 9.63%.
Properties
Rosettes from aq ethanol and chloroform/ petr
ether, mp 163-165°. uv max (95% ethanol): 252, 258, 262, 265, 269 nm (log ε 3.11, 3.08, 3.07, 3.01, 2.97).Derivative
Endiandric Acid C.
Nomenclature
CAS number: 76060-34-9
(1
R,1a
R,3
S,3a
R,6
S,6a
S,6b
R,7
S)-
rel-1,1a,2,3,3a,6,6a,6b-Octahydro-1-[(2
E,4
E)-5-phenyl-2,4-pentadienyl]-3,6-methanocyclobut[
cd]indene-7-carboxylic acid; 4-[(
E,E)-5′-phenylpenta-2′,4′-dien-1′-yl]tetracyclo[5,4,0,0
2,5,0
3,9]undec-10-ene-8-carboxylic acid.
C
23H
24O
2; mol wt 332.44.
C 83.10%, H 7.28%, O 9.63%.
Properties
Cryst from ethanol and methanol, mp 125-132° (variable and with dec). uv max (95% ethanol): 222, 228, 236, 280, 288 nm (log ε 4.10, 4.05, 3.89, 4.51, 4.53).