3600. Enviomycin
Nomenclature
Description and references
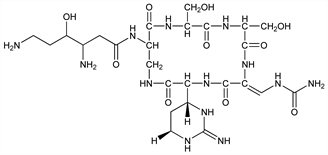
Polypeptide antibiotic produced by a mutant of Streptomyces griseoverticillatus var. tuberacticus. Isoln and characterization: T. Ando et al., J. Antibiot. 24, 680 (1971); A. Nagata et al., in Advan. Antimicrob. Antineopl. Chemother. vol. 1/2, M. Hejzlar et al., Eds. (University Park Press, Baltimore, 1972) pp 1039-1041. Prepn: J. Abe et al., DE 2133181; eidem, US 3892732 (1972, 1975 both to Toyo Brewing Co., Ltd.). Structural studies: H. Yoshioka et al., Tetrahedron Lett. 1971, 2043; T. Wakamiya, T. Shiba, J. Antibiot. 28, 292 (1975). Pharmacology: H. Hamakawa et al., Oyo Yakuri 8, 817 (1974), C.A. 82, 51530y (1975). Metabolism: T. Shimizu et al., Jpn. J. Antibiot. 27, 279 (1974). Total synthesis: T. Shiba et al., J. Antibiot. 32, 1078 (1979).
Derivative
Hydrochloride.Properties
White crystalline powder, mp >245° (dec). [α]D21 -19.1°. uv max (water, 0.1N HCl): 268 nm (E1%1cm 342); uv max (0.1N NaOH): 288 nm (E1%1cm 215). Very sol in water, slightly sol in methanol, ethanol. Insol in common organic solvents.Derivative
Sulfate.Nomenclature
Tuberactin (Toyo Jozo).Properties
White crystalline powder. Sol in water, slightly sol in usual organic solvents. LD50 in mice, rats (mg/kg): 485, 680 i.v. (Hamakawa).Therapeutic Category
Antibacterial (tuberculostatic).
Keywords
Antibacterial (Antibiotics); Polypeptides; Antibacterial (Tuberculostatic)