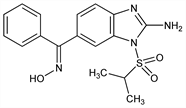
3601. Enviroxime
Nomenclature
CAS number: 72301-79-2
6-[(Hydroxyimino)phenylmethyl]-1-[(1-methylethyl)sulfonyl]-1H-benzimidazol-2-amine; (E)-2-amino-6-benzoyl-1-(isopropylsulfonyl)benzimidazole
oxime; anti-2-amino-1-(isopropylsulfonyl)-6-(α-hydroxyiminobenzyl)benzimidazole; LY-122772. C17H18N4O3S; mol wt 358.41.
C 56.97%, H 5.06%, N 15.63%, O 13.39%, S 8.95%.
Description and references
Benzimidazole deriv that inhibits rhinovirus multiplication. Prepn: C. J. Paget et al., DE 2638551; eidem, US 4118742 (1977, 1978 both to Lilly). Synthesis and sepn of syn and anti isomers: J. H. Wikel et al., J. Med. Chem. 23, 368 (1980). Inhibition of rhinovirus replication in organ culture: D. C. De Long, S. E. Reed, J. Infect. Dis. 141, 87 (1980). Metabolic studies: J. F. Quay et al., Fed. Proc. 39, 3079 (1980); C. J. Parli et al., ibid. 214. Activity vs rhinovirus infection in man: R. J. Phillpotts et al., Lancet 1, 1342 (1981). Prophylactic activity: F. G. Haden, J. M. Gwaltney, Antimicrob. Agents Chemother. 21, 892 (1982).