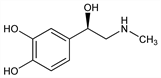
3619. Epinephrine
Nomenclature
Description and references
Endogenous catcholamine with combined α- and β-agonist activity. Principal sympathomimetic hormone produced by the adrenal medulla. Isoln from animal adrenal glands: Takamine, J. Soc. Chem. Ind. 20, 746 (1901); Aldrich, Am. J. Physiol. 5, 457 (1901). Synthesis of dl-form: Stolz, Ber. 37, 4149 (1904); Payne, Ind. Chem. 37, 523 (1961). Historic review of syntheses: Loewe, Arzneim.-Forsch. 4, 583 (1954). Resolution of dl-form: Fl"acher, Z. Physiol. Chem. 58, 189 (1908). Configuration: Pratesi et al., J. Chem. Soc. 1958, 2069. Acute toxicity: A. M. Lands et al., J. Pharmacol. Exp. Ther. 90, 110 (1947). HPLC determn in plasma and urine: C. R. Benedict, J. Chromatogr. 385, 369 (1987). Comprehensive description: D. H. Szulczewski, W.-H. Hong, Anal. Profiles Drug Subs. 7, 193-229 (1978). Physiologic review: Malmejac, Physiol. Rev. 44, 186 (1964). Review of biosynthesis: L. A. Pohorecky, R. J. Wurtman, Pharmacol. Rev. 23, 1-35 (1971); of pharmacology and clinical use in cardiopulmonary resuscitation: N. A. Paradis, E. M. Koscove, Ann. Emerg. Med. 19, 1288-1301 (1990); P. Hebert et al., J. Emerg. Med. 9, 487-495 (1991). Review of use in anaphylaxis: A. P. C. McLean-Tooke et al., Br. Med. J. 327, 1332-1335 (2003).
Properties
Minute crystals, gradually browning on exposure to light and air. mp 211-212°. mp ≈215° (dec) when rapidly heated. [α]D25 -50.0° to -53.5° (in 0.6N HCl). Very slightly sol in water, alc. Readily sol in aq solns of mineral acids, NaOH, and KOH. Insol in aq solns of ammonia and of the alkali carbonates. Insol in chloroform, ether, acetone, oils. LD50 i.p. in mice: 4 mg/kg (Lands).Derivative
Hydrochloride.Nomenclature
Derivative
d-Bitartrate.Nomenclature
Properties
Crystals, mp 147-154° (some dec). Darkens slowly on exposure to air and light. One gram dissolves in about 3 ml water. Slightly sol in alc.Derivative
dl-Form.Nomenclature
Properties
Sparingly sol in water, alcohol.Derivative
dl-Form hydrochloride.Nomenclature
Properties
Crystals from alcohol, mp 157°. Readily sol in water; sparingly sol in abs alc.Therapeutic Category
Bronchodilator; cardiostimulant; mydriatic; antiglaucoma.
Therapeutic Category (Veterinary)
Vasoconstrictor; cardiostimulant.Keywords
α-Adrenergic Agonist; β-Adrenergic Agonist; Antiglaucoma; Bronchodilator; Ephedrine Derivatives; Decongestant; Mydriatic