3620. Epiquinidine
Nomenclature
CAS number: 572-59-8
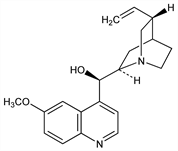
(9R)-6′-Methoxycinchonan-9-ol. C20H24N2O2; mol wt 324.42.
C 74.04%, H 7.46%, N 8.63%, O 9.86%.
Description and references
Occurs in cinchona bark. Isoln from quinoidine: Dirscherl, Thron, Ann. 521, 48 (1938). By epimerization of quinine or quinidine: Rabe, H"oter, J. Prakt. Chem. 154, 66 (1940). Stereochemistry: Prelog, Zalán, Helv. Chim. Acta 27, 535, 545 (1944); Prelog, H"afliger, ibid. 33, 2021 (1950); Roth, Pharmazie 16, 257 (1961). Synthesis: Grethe et al., Helv. Chim. Acta 55, 1044 (1972); Gutzwiller, Uskokovic, ibid. 56, 1494 (1973).
Properties
Lustrous leaflets from ether, mp 111-113°. [α]D25 +107.8° (c = 1.02 in ethanol). Freely sol in alcohol; moderately sol in ether. Shows more blue fluorescence in H2SO4 than quinidine or quinine. Forms a double sulfate with epiquinine.Derivative
Dihydrochloride.C20H24N2O2.2HCl; mol wt 397.34.
C 60.46%, H 6.60%, N 7.05%, O 8.05%, Cl 17.85%.
Properties
Crystals from alc, dec 195-196°. [α]D20 +46° (c = 0.8 in 99% alc).Derivative
Neutral dibenzoyl-d-tartrate.(C20H24N2O2)2.C18H14O8; mol wt 1007.13.
C 69.17%, H 6.20%, N 5.56%, O 19.06%.