3623. Epirubicin
Nomenclature
Description and references
Analog of the anthracycline antibiotic doxorubicin, q.v., differing only in the position of the C-4 hydroxy group of the sugar moiety. Prepn: F. Arcamone et al., DE 2510866; eidem, US 4058519 (1975, 1977 both to Soc. Farmaceut. Italia); eidem, J. Med. Chem. 18, 703 (1975); S. Penco, Process Biochem. 15(5), 12 (1980). Purification: E. Oppici et al., BE 898506; eidem, GB 2133005 (both 1984 to Farmitalia Carlo Erba). Comparison with doxorubicin of in vitro activity: A. Di Marco et al., Cancer Res. 36, 1962 (1976). Tissue distribution: C. Italia et al., Br. J. Cancer 47, 545 (1983); F. Arcamone et al., Cancer Chemother. Pharmacol. 12, 157 (1984). HPLC determn in biological fluids: P. E. Deesen, B. Leyland-Jones, Drug Metab. Dispos. 12, 9 (1984); G. Cassinelli et al., ibid. 506. Clinical trials in solid tumors: K. Kolaric et al., J. Cancer Res. Clin. Oncol. 106, 148 (1983). Review of activity in experimental tumors: A. Goldin et al., Invest. New Drugs 3, 3-21 (1985). Review of pharmacology and clinical efficacy: F. Ganzina, Cancer Treat. Rev. 10, 1-22 (1983); R. J. Cersosimo, W. K. Hong, J. Clin. Oncol. 4, 425-439 (1986); P. Hurteloup, F. Ganzina, Drugs Exp. Clin. Res. 13, 233-246 (1986). Review of epirubicin and other anthracycline antineoplastics: R. B. Weiss et al., Cancer Chemother. Pharmacol. 18, 185-97 (1986).
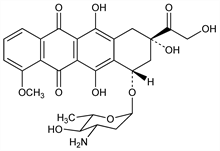
Derivative
Hydrochloride.Nomenclature
Properties
Red-orange crystals, mp 185° (dec). [α]D20 +274° (c = 0.01 in methanol). Solution should be protected from sunlight.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs; Anthracyclines; Topoisomerase II Inhibitor