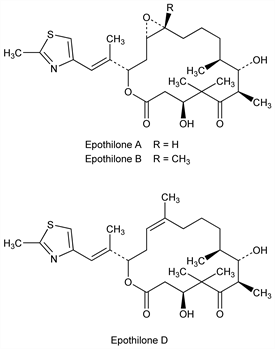
3628. Epothilones
Description and references
Microtubule stablilizing 16-membered macrolides which mimic the biological effects of paclitaxel, q.v., while retaining multidrug-resistant activity. Structurally lacking the taxane ring. Isoln of A and B from the myxobacterium Sorangium cellulosum: G. H"ofle et al., DE 4138042, (1993 to Ges. Biotech. Forsch.), C.A. 120, 52841 (1994). Purification of A and B, and mechanism of action: D. M. Bollag et al., Cancer Res. 55, 2325 (1995). Total synthesis of A: A. Balog et al., Angew. Chem. Int. Ed. 35, 2801 (1996); of B and D: D.-S. Su et al., ibid. 36, 757 (1997). Crystal structure of A and B, and conformation in soln: G. H"ofle et al., ibid. 35, 1567 (1996). Isoln and structural studies of 39 natural epothilone variants: I. H. Hardt et al., J. Nat. Prod. 64, 847 (2001). Isoln and crystallization of D: R. L. Arslanian et al., ibid. 65, 570 (2002). Effects of A and B on microtubule proteins in comparison with paclitaxel: R. J. Kowalski et al., J. Biol. Chem. 272, 2534 (1997). Review of properties and clinical potential: D. M. Bollag, Expert Opin. Invest. Drugs 6, 867-873 (1997); of biology, chemistry and therapeutic potential: K. C. Nicolaou et al., Angew. Chem. Int. Ed. 37, 2014-2045 (1998); idem et al., Chem. Commun. 2001, 1523-1535.