3635. Eprosartan
Nomenclature
Description and references
Prototype of the imidazoleacrylic acid angiotensin II receptor antagonists. Prepn: J. A. Finkelstein et al., EP 403159 (1990 to SmithKline Beckman); eidem, US 5185351 (1993 to SmithKline Beecham); J. Weinstock et al., J. Med. Chem. 34, 1514 (1991); R. M. Keenan et al., ibid. 36, 1880 (1993). Pharmacology: R. M. Edwards et al., J. Pharmacol. Exp. Ther. 260, 175 (1992). Receptor binding study: H. T. Schambye et al., Mol. Pharmacol. 47, 425 (1995). Clinical pharmacokinetics: D. Tenero et al., Biopharm. Drug Dispos. 19, 351 (1998). Clinical trial in hypertension: W. J. Elliott et al., J. Hum. Hypertens. 13, 413 (1999). Review of clinical experience: L. Ruilope, B. J"ager, Expert Opin. Pharmacother. 4, 107-114 (2003).
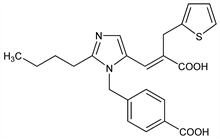
Properties
Crystals from methanol, mp 260-261°.Derivative
Monomethanesulfonate.Nomenclature
Properties
White to off-white crystalline powder, mp 248-250°. Insol in water. Freely sol in ethanol.Therapeutic Category
Antihypertensive.
Keywords
Angiotensin II Receptor Antagonist; Antihypertensive; Imidazoleacrylic Acid Derivatives