3650. Ergocryptine
Nomenclature
Ergokryptine.C32H41N5O5; mol wt 575.70.
C 66.76%, H 7.18%, N 12.16%, O 13.90%.
Description and references
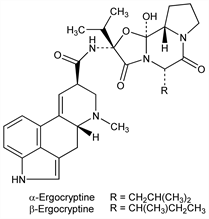
Two closely related isomers of the ergotoxine group which differ in the peptide portion of the molecule; α-ergocryptine yielding l-leucine upon hydrolysis, β-ergocryptine yielding l-isoleucine. The ergocryptine discussed in the literature prior to 1967 is now referred to as α-ergocryptine. Isoln from ergot: Stoll, Hofmann, Helv. Chim. Acta 26, 1570 (1943). Structure: Stoll et al., ibid. 34, 1544 (1951). Separation and purification: Stoll, Hofmann, US 2447214 (1948 to Sandoz). Separation of β-ergocryptine from α-ergocryptine: Schlientz et al., Experientia 23, 991 (1967); see also eidem, Pharm. Acta Helv. 43, 497 (1968). Synthesis of α- and β-ergocryptine: Stadler et al., Helv. Chim. Acta 52, 1549 (1969).
Derivative
α-Ergocryptine.Nomenclature
CAS number: 511-09-1
(5′α)-12′-Hydroxy-2′-(1-methylethyl)-5′-(2-methylpropyl)ergotaman-3′,6′,18-trione. Properties
Solvated prisms from acetone, benzene, methanol. With MeOH of crystn, mp 212° (dec). [α]D20 -120° (pyridine); -198° (chloroform). uv max (methanol): 241, 312.5 nm (log ε 4.31, 3.95). Freely sol in alcohol, chlorofom. Almost insol in water.Derivative
β-Ergocryptine.Nomenclature
CAS number: 20315-46-2
[5′α(S)]-12′-Hydroxy-2′-(1-methylethyl)-5′-(1-methylpropyl)ergotaman-3′,6′,18-trione. Properties
Rectangular plates from benzene, mp 173° (dec). [α]D20 -98° (c = 0.5 in pyridine); -179° (c = 0.5 in chloroform). uv max (methanol): 312 (log ε 3.93).Therapeutic Category
See Ergot.
Keywords
Antimigraine