3649. Ergocristine
Nomenclature
CAS number: 511-08-0
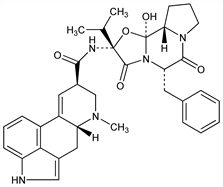
(5′α)-12′-Hydroxy-2′-(1-methylethyl)-5′-(phenylmethyl)ergotaman-3′,6′,18-trione. C35H39N5O5; mol wt 609.71.
C 68.95%, H 6.45%, N 11.49%, O 13.12%.
Description and references
Natural ergot alkaloid derived from lysergic acid; a member of the ergotoxine group. Isoln from ergot: Stoll, Burckhardt, Z. Physiol. Chem. 250, 1 (1937); Stoll, Hofmann, Helv. Chim. Acta 26, 1570 (1943). Structure: Stoll et al., ibid. 34, 1544 (1951). Separation and purification: Stoll, Hofmann, US 2447214 (1948 to Sandoz). Synthesis: Stadler et al., Helv. Chim. Acta 52, 1549 (1969).
Properties
Orthorhombic crystals with 2C6H6 from benzene. mp 155-157° (dec) (solvent-free base). [α]D20 -183° (chloroform). Very sol in ethyl and methyl alcohol, acetone, chloroform, ethyl acetate. Slightly sol in ether. Practically insol in water, petr ether.Derivative
Phosphate.Nomenclature
CAS number: 6424-36-8
Properties
Crystals, dec 195°.Derivative
Ethanesulfonate.Nomenclature
CAS number: 6055-56-7
Properties
Crystals, dec 207°.Derivative
8α-isomer.Nomenclature
CAS number: 511-07-9
Ergocristinine.