3652. Ergoflavin
Nomenclature
CAS number: 3101-51-7
(1S,1′S,3S,3′S,4S,4′S,4aS,4′aS,9aR,9′aR)-1,1′,3,3′,4,4′,9a,9′a-Octahydro-4,4′,8,8′,9a,9′a-hexahydroxy-3,3′-dimethyl-[7,7′-bi-1,4a-(epoxymethano)-4aH-xanthene]-9,9′,11,11′(2H,2′H)-tetrone; ergochrome CC(2,2′). C30H26O14; mol wt 610.52.
C 59.02%, H 4.29%, O 36.69%.
Description and references
Principal pigment from ergot: Freeborn, Pharm. J. 88, 568 (1912); Eglinton et al., J. Chem. Soc. 1958, 1833. Structure: McPhail et al., ibid. 1966, Sect. B, 18. Review of ergoflavin and other ergochromes: Franck, Flasch in Fortschr. Chem. Org. Naturst. 30, 151-206 (1973).
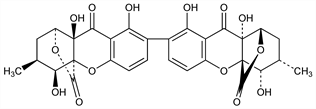
Properties
Yellow needles from methanol, decomp 350°. [α]D21 +37.5° (c = 1.236 in acetone). uv max: 240, 260, 381 nm (E1%1cm 350, 346, 130). Sol in acetone, pyridine; moderately sol in methanol, alcohol, ethyl acetate, dioxane; sparingly sol in ether, benzene. Practically insol in 2N aq NaHCO3.Derivative
Hexaacetate.C42H38O20; mol wt 862.74.
C 58.47%, H 4.44%, O 37.09%.