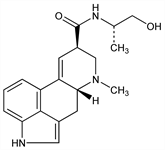
3655. Ergonovine
Nomenclature
Description and references
From some ergots: A. Stoll et al., US 2809920 (1957 to Saul & Co.). Prepn from d-lysergic acid and l(+)-2-amino-1-propanol: A. Stoll, A. Hofmann, Helv. Chim. Acta 26, 944 (1943); Pioch, US 2736728 (1956 to Lilly); Patelli, Bernardi, US 3141887 (1964 to Farmitalia). Structure: A. Stoll et al., Helv. Chim. Acta 34, 1544 (1951). Total synthesis: E. C. Kornfeld et al., J. Am. Chem. Soc. 78, 3087 (1956). Metabolism: Slaytor, Wright, J. Med. Pharm. Chem. 5, 483 (1962). Comprehensive description of the maleate: V. D. Reif, Anal. Profiles Drug Subs. 11, 273-312 (1982). Toxicity data: R. P. Beliles, Toxicol. Appl. Pharmacol. 23, 537 (1972). Clinical use in diagnosis of angina: L. A. DiCarlo, Jr. et al., Am. J. Cardiol. 54, 744 (1984); R. Nordlander, R. Orinius, Acta Med. Scand. 221, 47 (1987).
Properties
Tetrahedra from ethyl acetate, fine needles from benzene. Tends to form solvated crystals, mp 162°. [α]D20 +90° (in water). pK 6.8. Freely sol in lower alcohols, ethyl acetate, acetone; more sol in water than the other principal alkaloids of ergot; slightly sol in chloroform.Derivative
Hydrochloride.Properties
Needles from ethyl alcohol, dec 246°. [α]D25 +63° (c = 0.9). More sol in water than the hydrobromide.Derivative
Maleate.Nomenclature
Properties
Crystals, dec 167°. [α]D25 +48 to +57°. One gram dissolves in 36 ml water, 120 ml alcohol. Nearly insol in ether and chloroform. LD50 i.v. in mice: 8.26 mg/kg (Beliles).Derivative
Tartrate hydrate.Nomenclature
Basergin; Neofemergen.Properties
Crystals, slightly sol in water.Derivative
Hydracrylate.Nomenclature
Ergotrate-H (Lilly).Therapeutic Category
Oxytocic.
Therapeutic Category (Veterinary)
Oxytocic.Keywords
Oxytocic