3656. Ergosine
Nomenclature
CAS number: 561-94-4
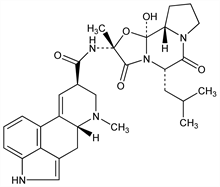
12′-Hydroxy-2′-methyl-5′α-(2-methylpropyl)ergotaman-3′,6′,18-trione. C30H37N5O5; mol wt 547.65.
C 65.79%, H 6.81%, N 12.79%, O 14.61%.
Description and references
Isoln of ergosine and ergosinine from ergot: Smith, Timmis, J. Chem. Soc. 1937, 396. Structure: Stoll et al., Helv. Chim. Acta 34, 1544 (1951). Stereochemistry: Stoll et al., ibid. 37, 2039 (1954). Total synthesis of ergosine and ergosinine: Stadler et al., ibid. 47, 1911 (1964).
Properties
Prisms from ethyl acetate, dec 228°. [α]D20 -161° (chloroform). Sol in chloroform; fairly sol in methanol, acetone; sparingly sol in ethyl acetate, benzene.Derivative
8α-Isomer.Nomenclature
Ergosinine; ergoclavinine.C30H37N5O5; mol wt 547.65.
C 65.79%, H 6.81%, N 12.79%, O 14.61%.