3690. d-Erythrose
Nomenclature
CAS number: 583-50-6
(2R,3R)-2,3,4-Trihydroxybutanal. C4H8O4; mol wt 120.10.
C 40.00%, H 6.71%, O 53.29%.
Description and references
From calcium d-arabonate by oxidation with H2O2: Ruff, Ber. 32, 3674 (1899); 33, 1799 (1900). From d-glucose: Perlin, Methods Carbohydr. Chem. 1 (Academic Press, New York, 1962) p 64. Synthesis of dl-erythrose: Sonogashira, Nakagawa, Bull. Chem. Soc. Jpn. 45, 2616 (1972).
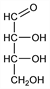
Properties
Syrup. Shows mutarotation. [α]D20 +1° → -14.5° (3 days, c = 11). Sol in water. Slowly reduces cold Fehling's soln. Sodium amalgam reduces it to natural, inactive erythritol. No aldehyde reaction with benzenesulfhydroxamic acid. Not fermented by yeast.Derivative
Phenylosazone.C16H18N4O2; mol wt 298.34.
C 64.41%, H 6.08%, N 18.78%, O 10.73%.