3691. l-Erythrose
Nomenclature
CAS number: 533-49-3
(2S,3S)-2,3,4-Trihydroxybutanal. C4H8O4; mol wt 120.10.
C 40.00%, H 6.71%, O 53.29%.
Description and references
From calcium l-arabonate by oxidation with H2O2: Ruff, Meusser, Ber. 34, 1365 (1901). From l-arabinose oxime: Wohl, Ber. 32, 3667 (1899). From l-arabonamide by treatment with alkaline NaOCl: Weerman, Rec. Trav. Chim. 37, 35 (1918).
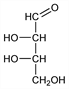
Properties
Syrup. Sweet taste. Shows mutarotation. [α]D24 +11.5° (8 min) → +15.2° (120 min) → +30.5° (final, c = 3): Felton, Freudenberg, J. Am. Chem. Soc. 57, 1640 (1935). Soluble in water. Heating with HCl yields lactic acid. Oxidation with Br converts it to l-erythronic acid. Reduces Fehling's soln slowly in the cold. Not fermented by yeast.Derivative
Phenylosazone.C16H18N4O2; mol wt 298.34.
C 64.41%, H 6.08%, N 18.78%, O 10.73%.