3698. Esculin
Nomenclature
CAS number: 531-75-9
6-(β-d-Glucopyranosyloxy)-7-hydroxy-2H-1-benzopyran-2-one; 6,7-dihydroxycoumarin 6-glucoside; esculoside; bicolorin; enallachrome; polychrome; Escosyl. C15H16O9; mol wt 340.28.
C 52.94%, H 4.74%, O 42.32%.
Description and references
In leaves and bark of horse chestnut tree Aesculus hippocastanum L., Hippocastanaceae. Extraction procedure: Tumann, Chem. Zentralbl. 1916, I, 1277. Synthesis: Merz, Hagemann, Naturwissenschaften 29, 650 (1941); eidem, Arch. Pharm. 282, 79 (1944); Amiard, Nominé, Bull. Soc. Chim. Fr. 1948, 476. Use in bile-esculin test for identification of group D streptococci: C. Chuard, L. B. Reller, J. Clin. Microbiol. 36, 1135 (1998).
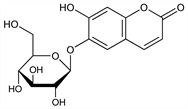
Derivative
Sesquihydrate.Properties
Needles from hot water, mp 204-206°. One gram dissolves in 580 ml water, 13 ml boiling water. Sol in hot alcohol, methanol, pyridine, ethyl acetate, acetic acid. Aq solns show blue fluorescence above pH 5.8. [α]D18 -78.4° (c = 2.5 in 50% dioxane). Absorption spectrum: Merz, Arch. Pharm. 270, 482 (1932); Goodwin, Pollock, Arch. Biochem. Biophys. 49, 1 (1954). Has vitamin P activity.Derivative
Pentaacetate dihydrate.Properties
Needles from methanol, mp 163-164°.Derivative
4-Methylesculin.C16H18O9; mol wt 354.31.
C 54.24%, H 5.12%, O 40.64%.
Description and references
Prepn: Velluz, Amiard, Bull. Soc. Chim. Fr. 1948, 1109.