3699. Eseridine
Nomenclature
CAS number: 25573-43-7
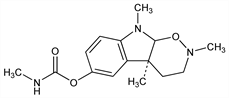
(4aS-cis)-2,3,4,4a,9,9a-Hexahydro-2,4a,9-trimethyl-1,2-oxazino[6,5-b]indol-6-ol methylcarbamate ester; physostigmine aminoxide; eserine aminoxide; eserine oxide. C15H21N3O3; mol wt 291.35.
C 61.84%, H 7.27%, N 14.42%, O 16.47%.
Description and references
From Calabar bean (Physostigma venenosum Balf., Leguminosae): Polonovski, Nitzberg, Bull. Soc. Chim. Fr. [5] 17, 244 (1915); 21, 191 (1917). Structure: Stedman, Barger, J. Chem. Soc. 127, 247 (1925). Revised structure: Hootele, Tetrahedron Lett. 1969, 2713. Stereochemistry: Robinson, Moorcroft, J. Chem. Soc. C 1970, 2077. Total synthesis of racemate: K. Shishido et al., Chem. Commun. 1986, 904.
Properties
Rectangular prisms from ether, mp 129°. [α]D15 -175° (alc). Weak base, just alkaline to litmus; does not form crystn salts with mineral acids. Almost insol in water. Sol in alc, chloroform, benzene, ether, petr ether, acetone, dil acids.Derivative
Salicylate.Nomenclature
CAS number: 5995-96-0
Génésérine 3 (Amido). C22H27N3O6; mol wt 429.47.
C 61.53%, H 6.34%, N 9.78%, O 22.35%.
Therapeutic Category
Cholinergic.
Keywords
Cholinergic