3700. Esmolol
Nomenclature
Description and references
Cardioselective β-adrenergic blocker. Prepn: E. I. Carlsson et al., EP 41491 (1982 to A.B. H"assle), C.A. 96, 122391f (1982); P. W. Erhardt et al., EP 53435; eidem, US 4593119 (1982, 1986 both to Am. Hosp. Supply); P. W. Erhardt et al., J. Med. Chem. 25, 1408 (1982). Pharmacology: R. J. Gorczynski et al., J. Cardiovasc. Pharmacol. 5, 668 (1983); eidem, ibid. 6, 1548 (1984). Pharmacokinetics: A. Yacobi et al., J. Pharm. Sci. 72, 711 (1983). GC-MS determn in blood: C. Y. Sum, A. Yacobi, ibid. 73, 1177 (1984). Clinical studies of effect in supraventricular tachycardia: G. Klein et al., Int. J. Clin. Pharmacol. Ther. Toxicol. 22, 112 (1984); R. J. Gray et al., J. Am. Coll. Cardiol. 5, 1451 (1985). Symposium on pharmacology and clinical efficacy: Am. J. Cardiol. 56(11), 1F-62F (1985). Review of pharmacology and clinical efficacy: P. Benfield, E. M. Sorkin, Drugs 33, 392-412 (1987).
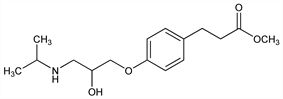
Properties
Oil, gradually forming crystalline rosettes at room temp, mp 48-50°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from methanol-ether, mp 85-86°. Very sol in water; freely sol in acetone. Partition coefficient (octanol/water, pH 7.0): 0.42.Therapeutic Category
Antiarrhythmic.
Therapeutic Category (Veterinary)
Antiarrhythmic.Keywords
β-Adrenergic Blocker; Antiarrhythmic