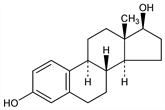
3703. Estradiol
Nomenclature
Description and references
Potent mammalian estrogenic hormone produced by the ovary. Triggers the production of gonadotropins leading to ovulation. Isoln from follicular fluid of sow ovaries: D. W. MacCorquodale et al., J. Biol. Chem. 115, 435 (1936). Has also been isolated from urine of pregnant women and mares. Prepn from estrone: A. Butenandt, C. Georgens, Z. Physiol. Chem. 248, 129 (1937); F. Hildebrandt et al., US 2096744 (1938 to Schering); from cholesterol: H. H. Inhoffen, G. Zühlsdorff, Ber. 74, 1914 (1941). Total syntheses: U. Eder et al., Ber. 109, 2948 (1976); W. Oppolzer, D. A. Roberts, Helv. Chim. Acta 63, 1703 (1980). Pharmacology: R. W. Lievertz, Am. J. Obstet. Gynecol. 156, 1289 (1987). HPLC determn in plasma and urine: W. Slikker et al., J. Chromatogr. 224, 205 (1981). Pharmacokinetics: W. Kuhnz et al., Arzneim.-Forsch. 43, 966 (1993). Clinical evaluation in osteoporosis: J. W. W. Studd et al., Br. J. Obstet. Gynaecol. 101, 787 (1994). Comprehensive description: E. G. Salole, Anal. Profiles Drug Subs. 15, 283-318 (1986). Physiologic review: H. G. Burger, Int. J. Gynaecol. Obstet. 1989, Suppl. 1, 5-9. Review of clinical safety and efficacy in estrogen replacement therapy: A. Cheang et al., Drug Saf. 9, 365-379 (1993); H. L. Judd, J. Reprod. Med. 39, 343-352 (1994).
Properties
White crystalline powder. Prisms from 80% alc, stable in air, mp 173-179°. [α]D25 +76 to +83° (dioxane). uv max: 225, 280 nm. Precipitated by digitonin. Practically insol in water. Freely sol in alcohol; sol in acetone, dioxane, other organic solvents; solns of fixed alkali hydroxides; sparingly sol in vegetable oils. 1 mg = 10,000 international units.Derivative
Hemihydrate.Nomenclature
Derivative
3-Acetate.Nomenclature
Derivative
3-Benzoate.Nomenclature
Properties
Crystals from alc, mp 191-196°. Stable in air. [α]D25 +58 to +63° (c = 2 in dioxane). Sol in alc, acetone, dioxane; slightly sol in ether, vegetable oils.Derivative
17β-Cyclopentanepropanoate.Nomenclature
Description and references
Prepn: Ott, US 2611773 (1952 to Upjohn).
Properties
Crystals from benzene + petr ether, mp 151-152°. [α]D25 +45° (chloroform). Sol in ether, methanol, benzene, chloroform, peanut oil, cottonseed oil, corn oil, sesame oil.Derivative
Dipropionate.Nomenclature
Description and references
Prepn: K. Miescher, C. Scholz, US 2205627 (1940 to Ciba).
Properties
mp 104-105°.Derivative
17-Heptanoate.Nomenclature
Description and references
Prepn: Gauthier et al., Ann. Pharm. Fr. 16, 757 (1958).
Properties
Crystals from diisopropyl ether, mp 94-96°.Derivative
17-Valerate.Nomenclature
Description and references
Prepn: K. Miescher, C. Scholz, US 2233025 (1941 to Ciba). Comprehensive description: K. Florey, Anal. Profiles Drug Subs. 4, 192-208 (1975).
Properties
Crystals, mp 144-145°.Caution
These substances are listed as known human carcinogens: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-115.Therapeutic Category
Estrogen.
Therapeutic Category (Veterinary)
Estrogen.Keywords
Estrogen; Steroidal